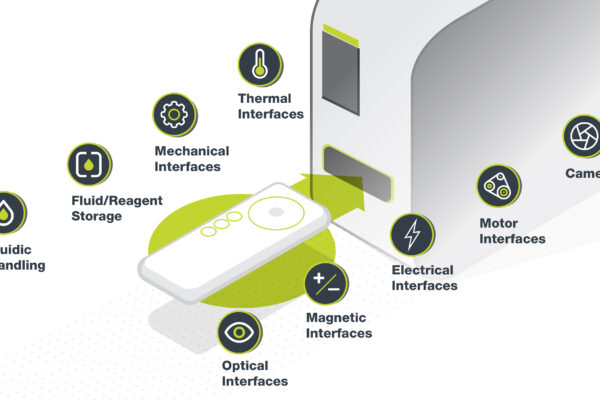
Quality Assurance
Key Tech’s quality assurance team supports companies, from startups to global corporations, to ensure that products and processes comply with the latest quality standards and regulations. Key Tech has over twenty-five years of hands-on experience developing medical, IVD, pharmaceutical, and life science devices under FDA and ISO compliant quality management systems. Our quality assurance consulting services are offered as both standalone engagements and as part of our technical programs.