End-To-End Product Development
Translating Your Product Development Needs into RealityOur Process
Our team works with you through every phase of our ISO 13485 compliant development process. Our carefully sequenced De-Risking, Development, and Commercialization phases ensure the right decisions are made at critical points in the development process.
Ideation
Requirements and Planning
The ideation process begins with understanding the needs of our clients, intended product users, and other stakeholders to ensure that both program constraints and product objectives are clearly understood and documented.
Learn more about how Key Tech’s Product Conceptualization service, Voice of Customer (VOC) process, and Quality and Regulatory experts ensure you have well-defined user and market inputs, a sound development plan, and the material needed to generate excitement with stakeholders and investors.
Concept Exploration
We research, brainstorm, and generate solution concepts that balance performance, usability, and stakeholder priorities—often leading to valuable new IP. Our team focuses on defining the right architecture early through system block diagrams, space-claims, and use workflows to guide key decisions and reduce long-term costs. Risk management is built into every step, addressing technical and programmatic challenges before they impact downstream development.
Learn more about how Key Tech’s Architecture Development process provides confidence that you’re developing the right product with the right approach.
Feasibility
We derisk and validate concepts early to ensure your technology can be demonstrated within a realistic system architecture before significant investment. Our team stress-tests and refines initial designs to build a strong foundation, setting your program on a faster, more confident path to success.
Learn more about how Key Tech uses Test Bed Development and Human Factors Studies to collect the critical data for proving technical and product feasibility.
Development
Once the architecture is validated, your product moves into formal, design-controlled development. This phase typically includes Alpha and Beta prototyping with functional testing to confirm the integrated design meets user and technical requirements. Our shared goal: a fully tested product ready for manufacturing ramp-up and successful launch.
Alpha and Beta Development
Alpha and Beta development phases provide critical proof points for your leadership team and investors. In Alpha, we design, prototype, and test the integrated system to demonstrate market and technical viability. In Beta, we refine the design based on Alpha learnings and conduct further testing to ensure it meets all user and technical requirements. Rapid prototyping and low-volume manufacturing provide devices for technical characterization, gathering client feedback, and human factors studies.
Commercialization
Your end goal is to commercialize a successful product. We will prepare a production-ready design package and manufacture initial builds at Key Tech or work closely with another manufacturing team to ensure a smooth ramp-up.
Pilot Builds
As part of our Manufacturing Solutions, Key Tech rapidly builds in-house pilot units suitable for clinical studies, electrical safety testing, and final verification and validation. These builds use production materials and follow defined pilot assembly and checkout procedures.
We also support alternate manufacturing paths—collaborating with client in-house teams or vetted contract manufacturers. Key Tech maintains design ownership through pilot builds and ramp-up to ensure continuity, reduce risk, and accelerate your path to market.
Verification and Validation
Our Systems Engineering teams verify that hardware, software, firmware, and system outputs meet defined input requirements. We also coordinate with accredited labs to complete EMC, electrical safety, biocompatibility, and other compliance testing.
In parallel, we work closely with your team to validate that the finished product meets user needs, conducting summative usability testing to confirm that safety-related risks are fully addressed.
Regulatory Approvals
Our Quality & Regulatory team will guide your product through the necessary stage gates to obtain market approval. At this point, we will be confident that the product design and supporting DHF are in compliance with all intended sales markets, allowing you to focus energy on product sales and marketing activities.
Manufacturing Ramp Up
Once the product is approved and the design is complete, Key Tech accelerates the transition to high-quality, high-volume production. We embed manufacturing design principles throughout development to reduce total product cost while maximizing reliability and manufacturability. Our engineers support scale-up by developing custom assembly and quality control procedures, along with software and fixtures that streamline production, increase throughput, and maintain quality at speed.
On Market Improvements
On-market improvements can strain your supply chain and manufacturing systems. Key Tech provides on-demand, multidisciplinary support to enhance launched products within existing production constraints. We also identify and implement design updates—from new features to hardware or software enhancements—when increased functionality or reliability is needed.
Resources
Want to learn more about how all of this works? Hear product development stories straight from clients and dig deeper into how our process can set you up for success.
Read
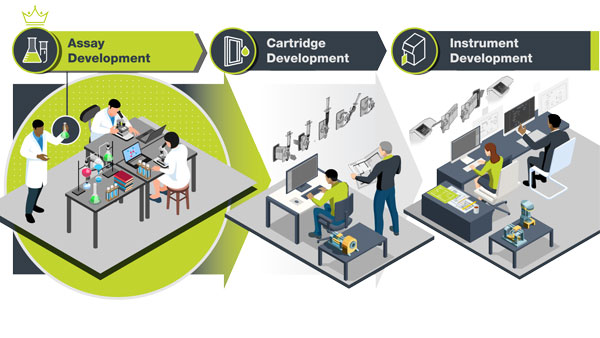
Benefits of IVD Cartridge and Instrument Development by One Partner
Save time, save money, prevent go-to and post-market challenges.
Learn How ›
Assay Is King
Assay performance should be the priority when developing complex cartridge and instrument systems.
Learn Why ›
Case Studies
Our end-to-end process transforms complex technologies into intuitive products for the world’s leading medical technology brands. Learn about some of our latest work on the market today.
Capabilities
Transforming complex technologies into intuitive experiences is no easy task. Offering an industry-leading, high-quality development process for various products, according to regulatory requirements, for multiple markets requires even more tenacity and an impeccable team. At Key Tech, our multidisciplinary team hails from many professional backgrounds. This widespread expertise allows us to work confidently, in compliance, across industries.