Medical Device
Get the right data, faster, to inform critical design decisions.
The quicker you can get the right data, through each stage of medical device development, the more likely your product will be a commercial success. But capturing and processing critical data is only the beginning. To make your device a success, you need to do it more efficiently than anyone else. And you have to do it with strict medical device safety and regulatory standards in mind.
That’s where Key Tech comes in. Our multidisciplinary medical device product development team has invented and developed some of the most complex – yet intuitive and easy to use – platforms on the market today. We focus on developing functional design solutions to get you the right data, faster.
Go with a process for success.
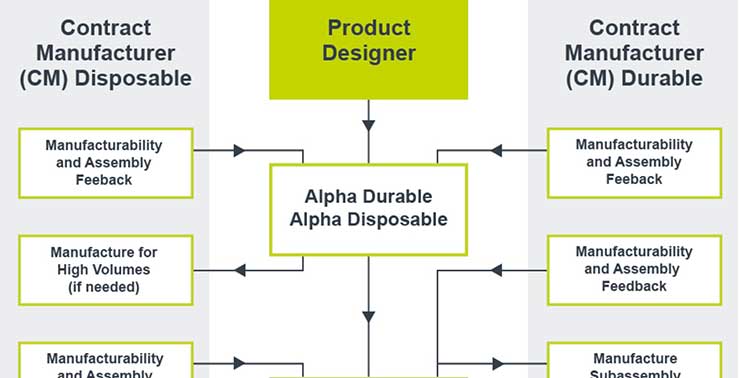
Our designers and engineers work collaboratively to carefully evaluate needs and develop system requirements, while assuring quality and managing risk.
Working across disciplines, we prioritize de-risking at each stage of design and development, to evaluate and validate architecture decisions. Further, our software development process follows the IEC 62304 standard whenever applicable, so the data you collect has that development pedigree behind it, giving you the confidence that the design is sound.
In medical device development, the right team is critical. We have over 24 years experience helping clients get the right data, faster. Let’s take the first step to long-term success, together.
Services for Medical
Device Development Groups
Independent
Technical Reviews
Early Concept Exploration –
User-Focused & Technical
Voice of Customer
Studies
Alpha and Beta
Prototype Development
Test Bed
Development

Relevant Work

In-Home Therapeutics
Migraine respiratory device
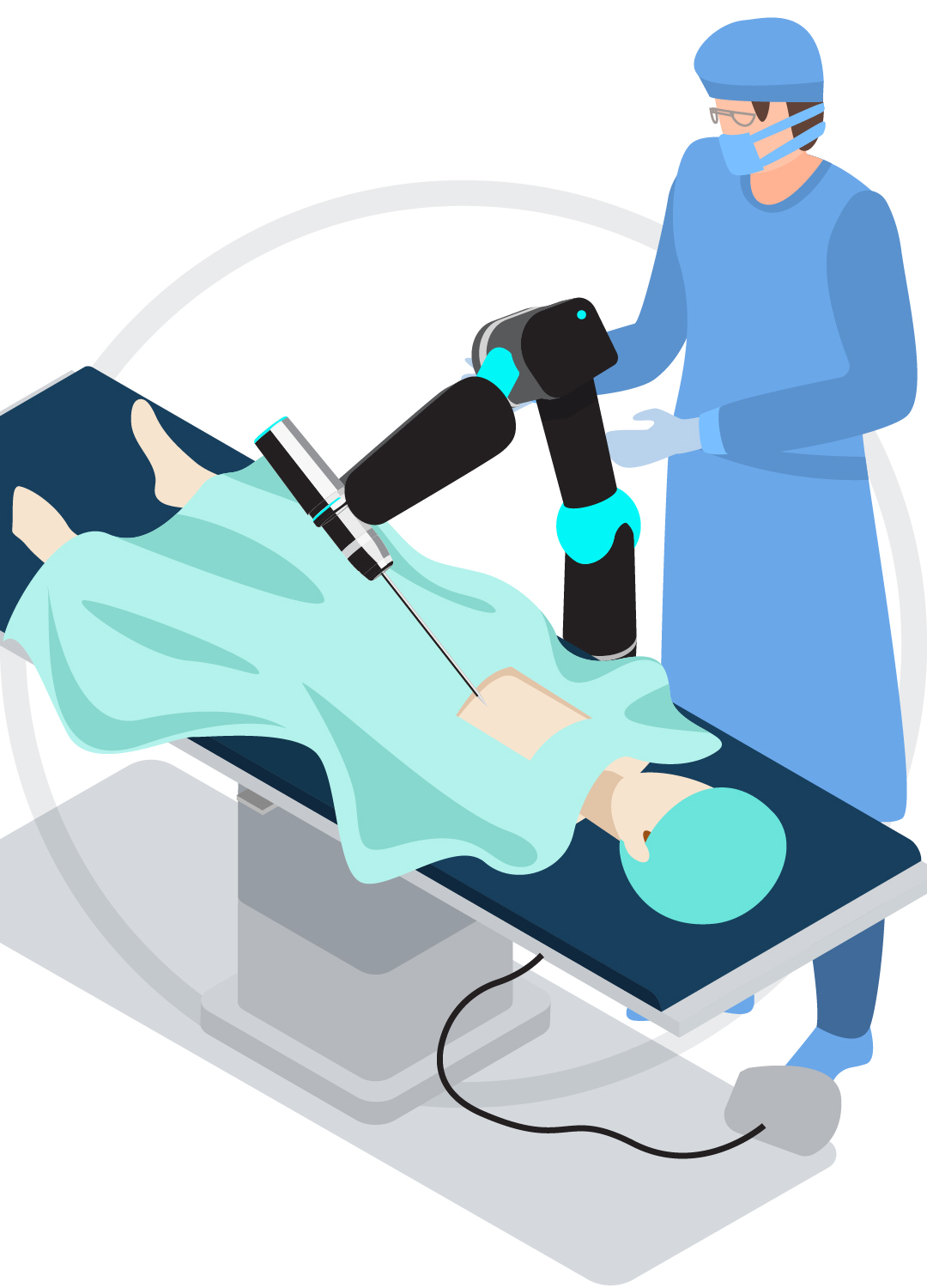
Surgical Robotics
Surgical positioning system
BioProcessing Platforms
Cell-based medicine processing system
Women’s Health
Laparascopic surgical tool
Wearables
On-body infusion pump

Bioelectronic Therapy
Wearable ultrasound device
Respiratory Care
Targeted temperature management system
Interventional Cardiology
Novel device ideation

Surgical Systems
Cart-based interventional products
Chronic Disease Management
Wearables and apps for diabetes care

Hospital Equipment
IV pumps and general anesthesia systems
Electrified Consumables
Smart consumables for active patient management