Speed is the key
At Key Tech, we believe that speed-to-data is a competitive advantage. We’ve worked in IVD consumable and instrument development for over 20 years, and we understand what it takes to create success in the marketplace.
Right now, in-home diagnostics and technologies like genetic sequencing and CRISPR are driving competition like never before. Older technologies are advancing; PCR tests are now faster and more accurate than ever, and antigen tests are becoming more specific. No matter what type of clinical situation, a new IVD platform must be reliable, accurate, and easy to use.
Solving the unsolvable.
Key Tech will ask the tough assay integration and automation questions to help you wade through the unknowns and find the right solution. We take pride in our reputation for proving the feasibility of complex technologies, then translating them to a viable commercial product.
We’ve formalized a framework to look at the whole system and ensure the instrument and consumable serve the assay. (See our Framework for Evaluating Assays for Automation.) By working closely with the client assay team, we are able to prove feasibility and burn down risk by building custom test fixtures that help inform product architecture decisions.
Our engineers and designers have successfully streamlined the process of IVD design and development for a wide range of customers like Thermo Fisher, Bio-Rad and GenMark Diagnostics (now a part of Roche). No matter what type of assay, we’ll help you deliver accurate clinical data with a competitive edge.
Services for In Vitro
Diagnostics Development Groups
Independent
Technical Reviews
Early Concept Exploration –
User-Focused & Technical
De-Risking and
Data Collection
Alpha and Beta
Prototype Development
Test Bed
Development
Relevant Work
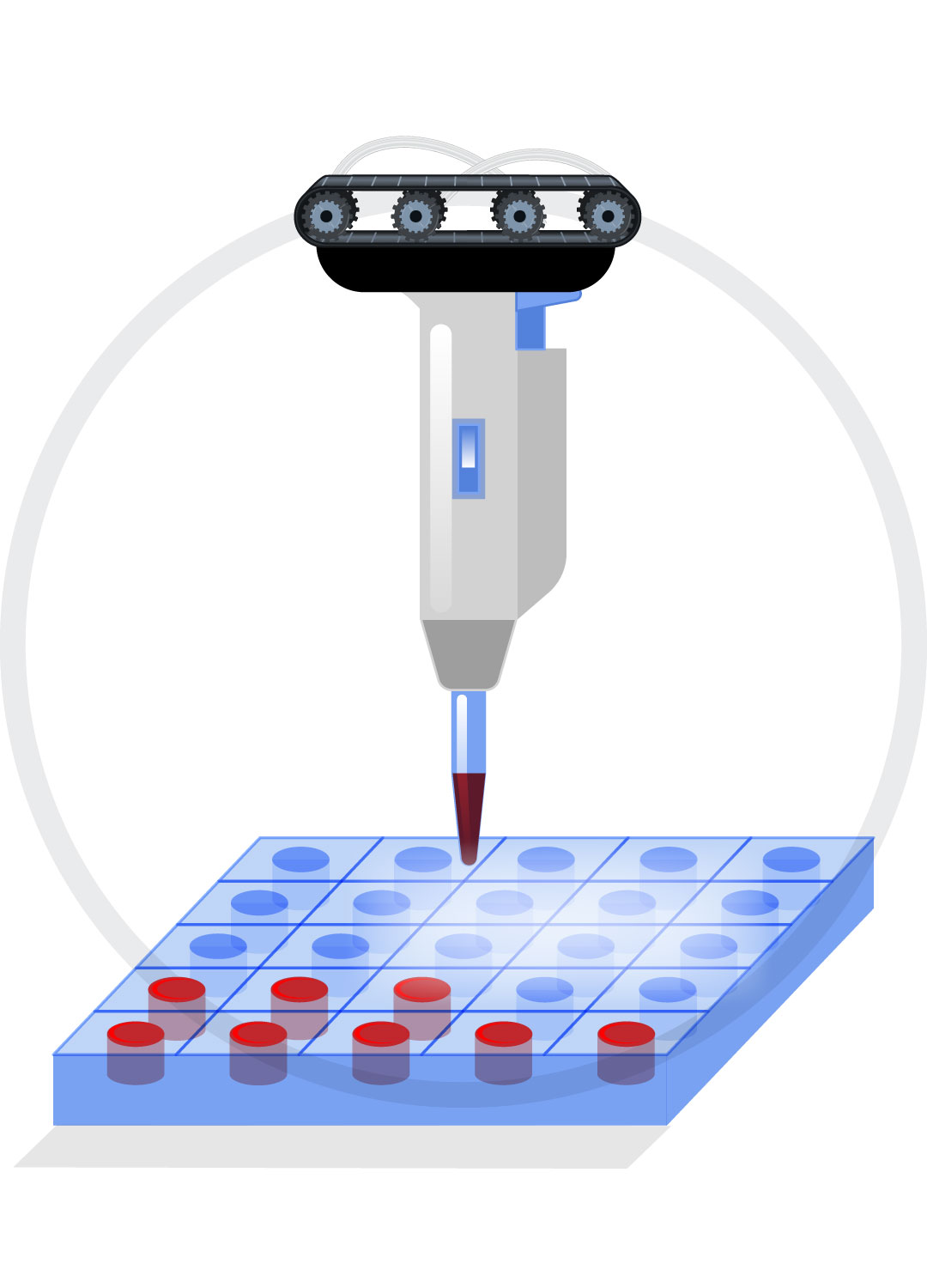
Liquid Handlers
Custom gantry and pipettor design

Sample Prep
Instruments
Purification platform

Sample to
Answer Platforms
Genetic and molecular multiplex instruments and consumables

Point of Care
Instruments
POC hematocrit meter

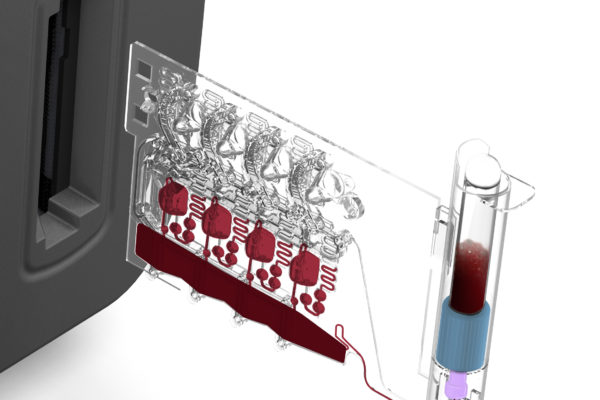
Cartridge Architecture & Design
Custom cartridge design to serve the assay

Onboard Reagents
Liquid, dried, pelleted, lyophilized
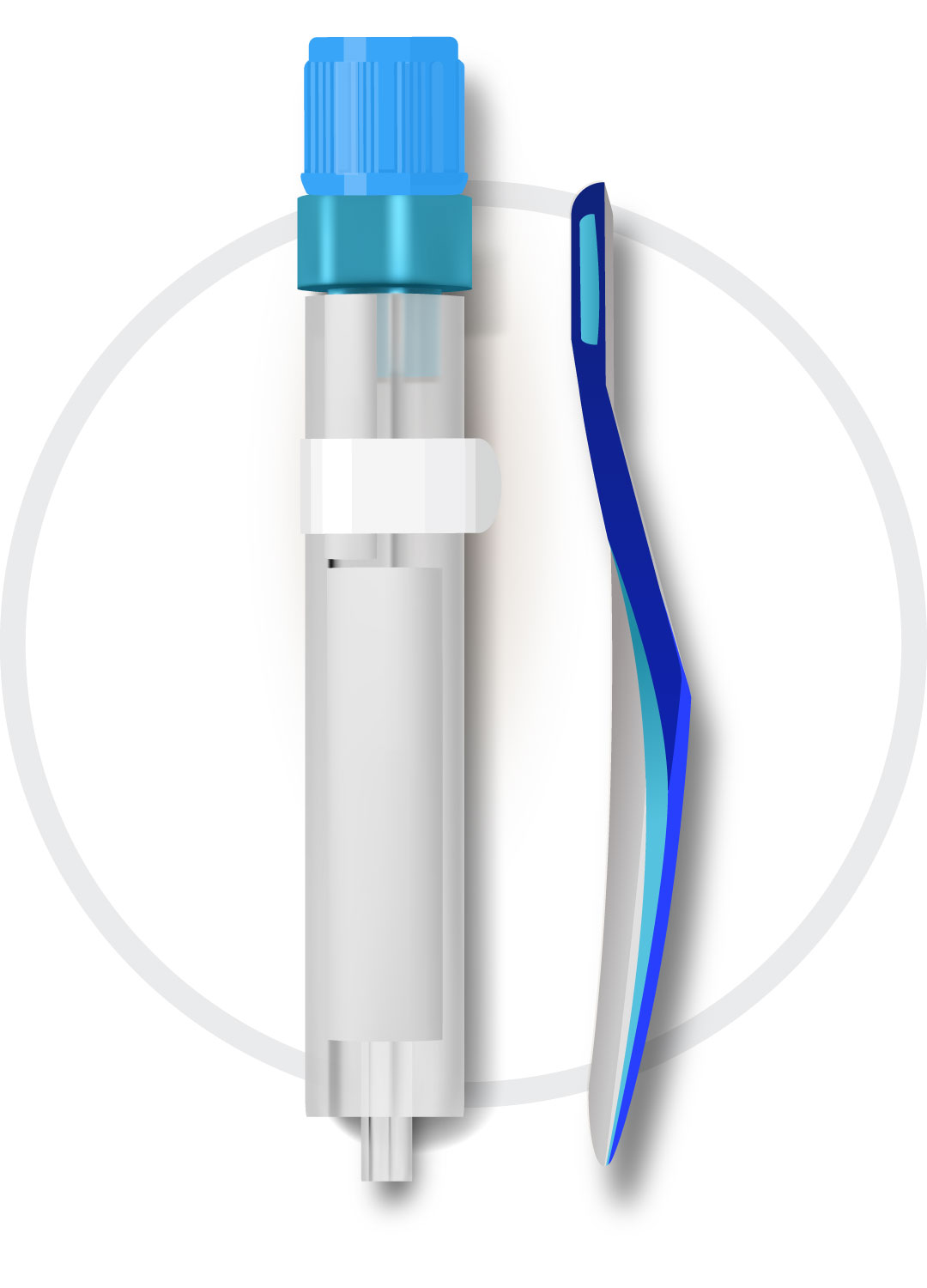
Sample Collection
Oral fluid collection device

Life Sciences
RUO technology, instruments, and consumables

Algorithm Development
Image detection, machine vision, precision detection algorithm

Detection Technologies
De-risking and integration of optical, ultrasonic, and electrical potential measurements

GUI & Workflow Definition
Design for ease of use

Test Fixtures
Smart test beds for R&D and verification
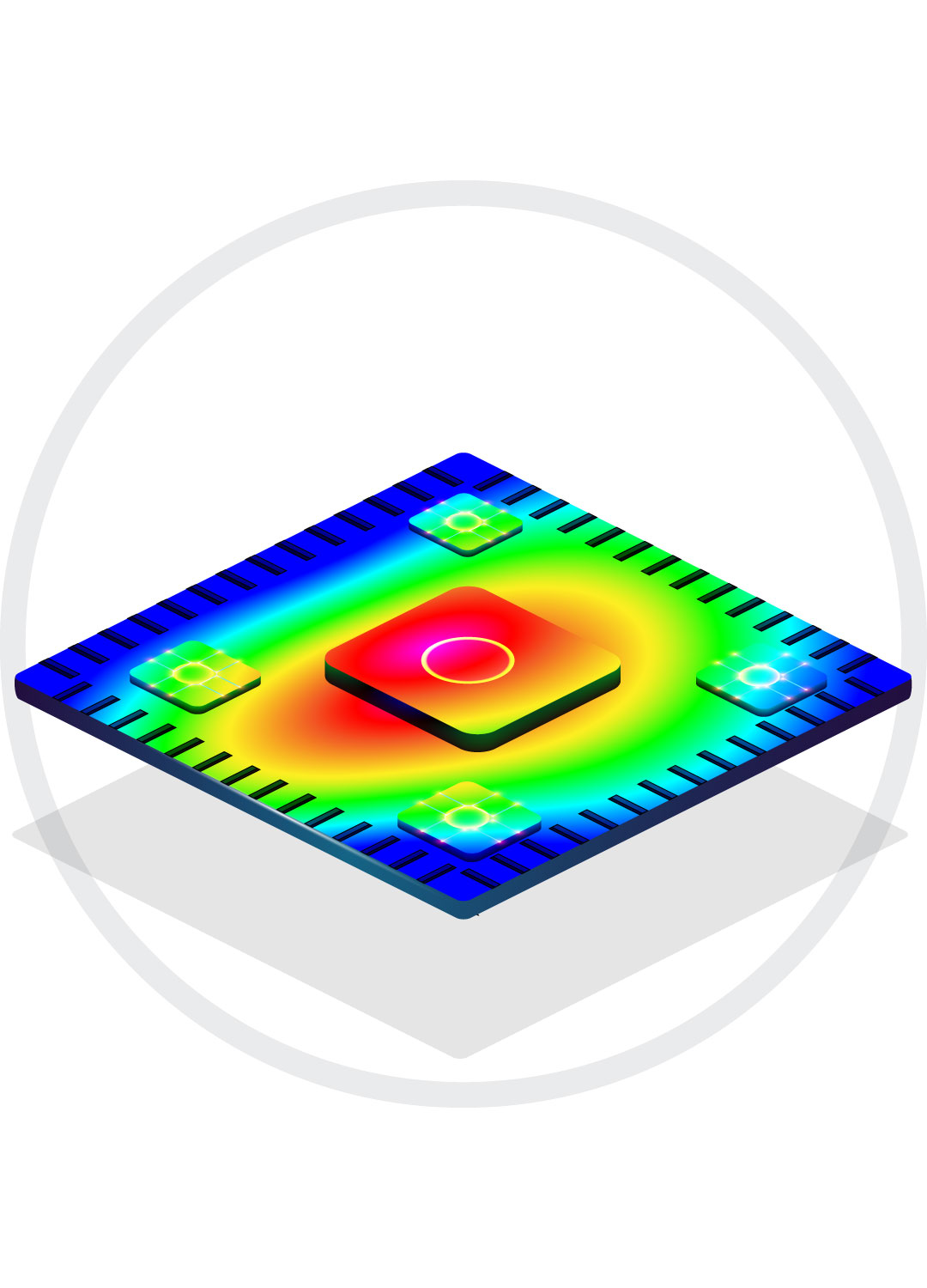
Thermal Control Management
Heating, interface design, materials, feedback and control
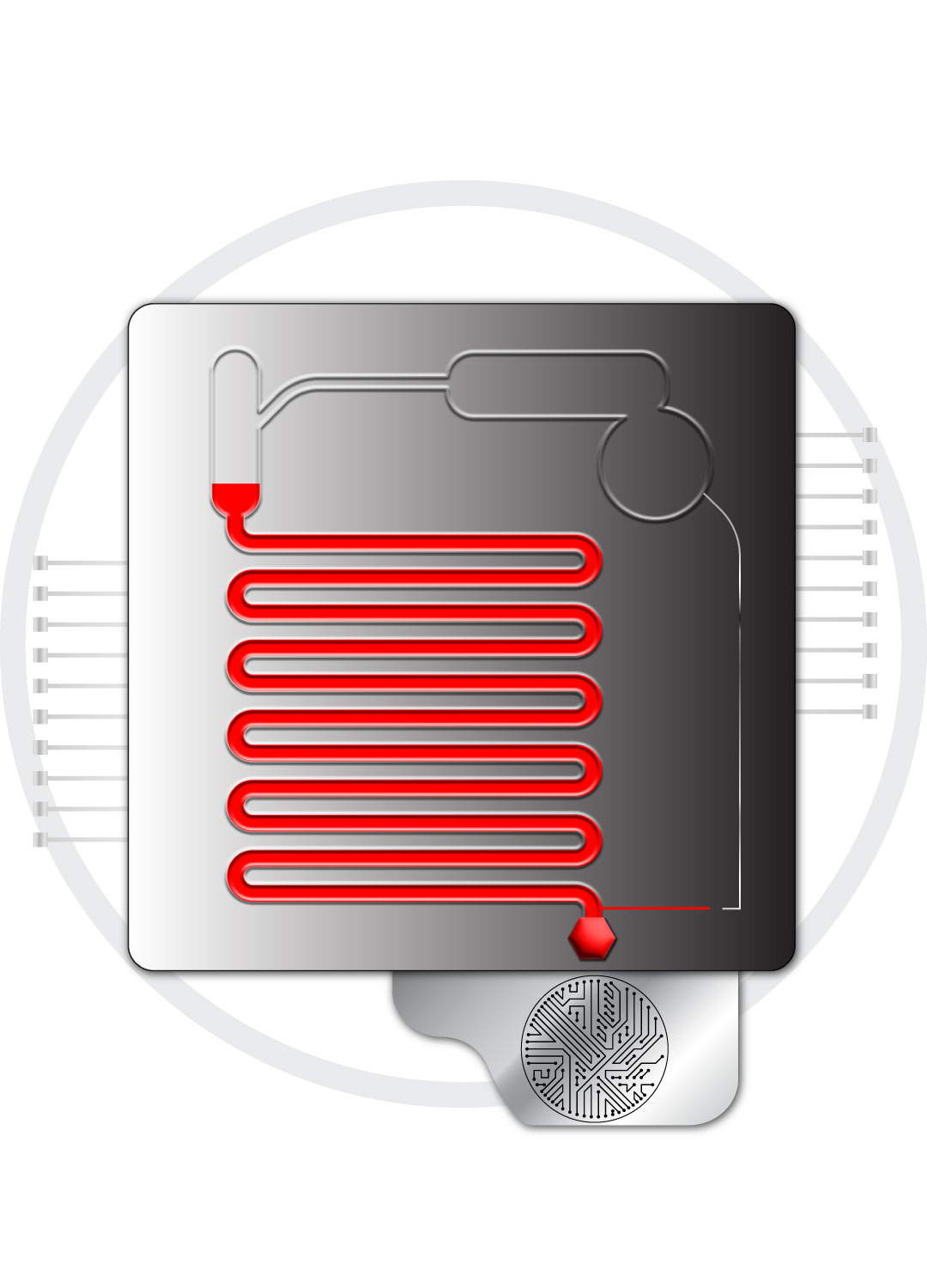
Fluidic Control
Fluidic motivation of various volumes and types of fluids
Novel Tech De-risking
Digital microfluidics

Complex Durable to Consumable Interface Design
Reliable actuation for various types of connections