Category
Computer Engineering, IVD, Mechanical Engineering, Software EngineeringAbout This Project
Autonomous Medical Devices Incorporated (AMDI)
Fast PCR
AMDI engaged Key Tech to conduct full scope product development of AMDI’s Fast PCR CLIA waived IVD instrument, for point of care respiratory panel testing. AMDI brought to the table a functional benchtop R&D fixture and proven assay-on-cartridge results to kick off the development program. Key Tech evolved this R&D fixture into a fully developed instrument, including all hardware and software, complete with a full fleet of Beta instruments to support clinical studies.
In the process of executing complex systems engineering and product development on a tight schedule, Key Tech incorporated the following notable achievements in this program:
- Custom optical engine design – Key Tech miniaturized AMDI’s optical sensing engine to allow multi-wavelength interrogation of their cartridge in a tight form factor without sacrificing signal quality
- Custom PCR mechanism design – Key Tech incorporated a novel cooling mechanism to AMDI’s PCR mechanism to push the limits on PCR speed, supporting a total sample-to-answer assay time of <10 minutes
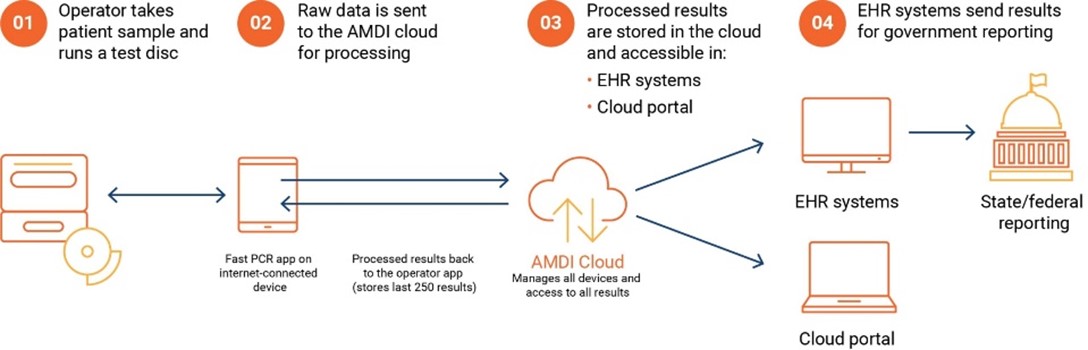
- Custom app-based UI – Key Tech developed a tablet and app-based UI for user interaction with the instrument and to coordinate instrument-to-cloud communication of instrument data
In addition to full scope design of the instrument and software, Key Tech manufactured full Alpha and Beta fleets of AMDI’s instrument. Alpha instruments were prioritized for usability testing and assay development. Beta instruments furthered assay development and ultimately were used at clinical studies.
The result of the partnership between AMDI and Key Tech is a 32-plex point of care system which can produce a respiratory diagnosis in <10 minutes, at a low price point to customers, in a small instrument size consistent with POC settings, and with an elegant and effortless user interface.
- Ep 44 The Real Cost of Adding Cybersecurity Late in Medical Device Development - February 4, 2026
- Ep 43 MedTech’s 11 Year Exit Problem— and What It Means for Raising Capital - December 12, 2025
- Ep 42 From Lab to Clinic: Building Safer Tools for Mothers and Babies - October 29, 2025