07 May Working with Client Assay Teams
You’ve worked tirelessly on the benchtop and developed a cutting edge assay that you know will make a big difference in the medical field. Now what?
Taking an assay from what works on the bench to what will work in a high-throughput diagnostic product is tough work. Developing a product that meets user and market needs means rethinking your priorities – where the focus for early assay development has been on accuracy and repeatability, new considerations like disposable/instrument cost, assay time, and disposable shelf life are key to designing a system that will be accepted by your potential customers. How can we balance these competing interests without sacrificing assay efficacy?
Establish achievable requirements.
While most assays are developed with precise benchtop lab equipment, in many cases this level of precision is not required for a successful assay. Unnecessarily precise control of fluids and temperatures can result in lengthy development schedules and drive instrument price beyond what is attractive for the market, so it is important to push the boundaries of your assay to understand sensitivities. Where a handheld pipettor might have a volumetric accuracy of better than ±1%, will the assay work with accuracies of ±10%? Where an off the shelf dry bath or thermocycler might report accuracies and uniformities of <±0.25C, is that level of thermal control necessary to get reliable results? Understanding the answers to these questions early in development is important for defining the range of viable instrument architectures.
Sensitivity studies for volumetric and thermal variability can be time-consuming to conduct on the benchtop, especially for longer and more complex assays. Often it is necessary to develop one or more test fixtures to expedite this testing. It is important for your assay team and design team to work together to understand the trade-offs of chemistry precision and product complexity, designing an appropriate set of experiments to reduce testing and development schedules.
Ruggedize your reagents.
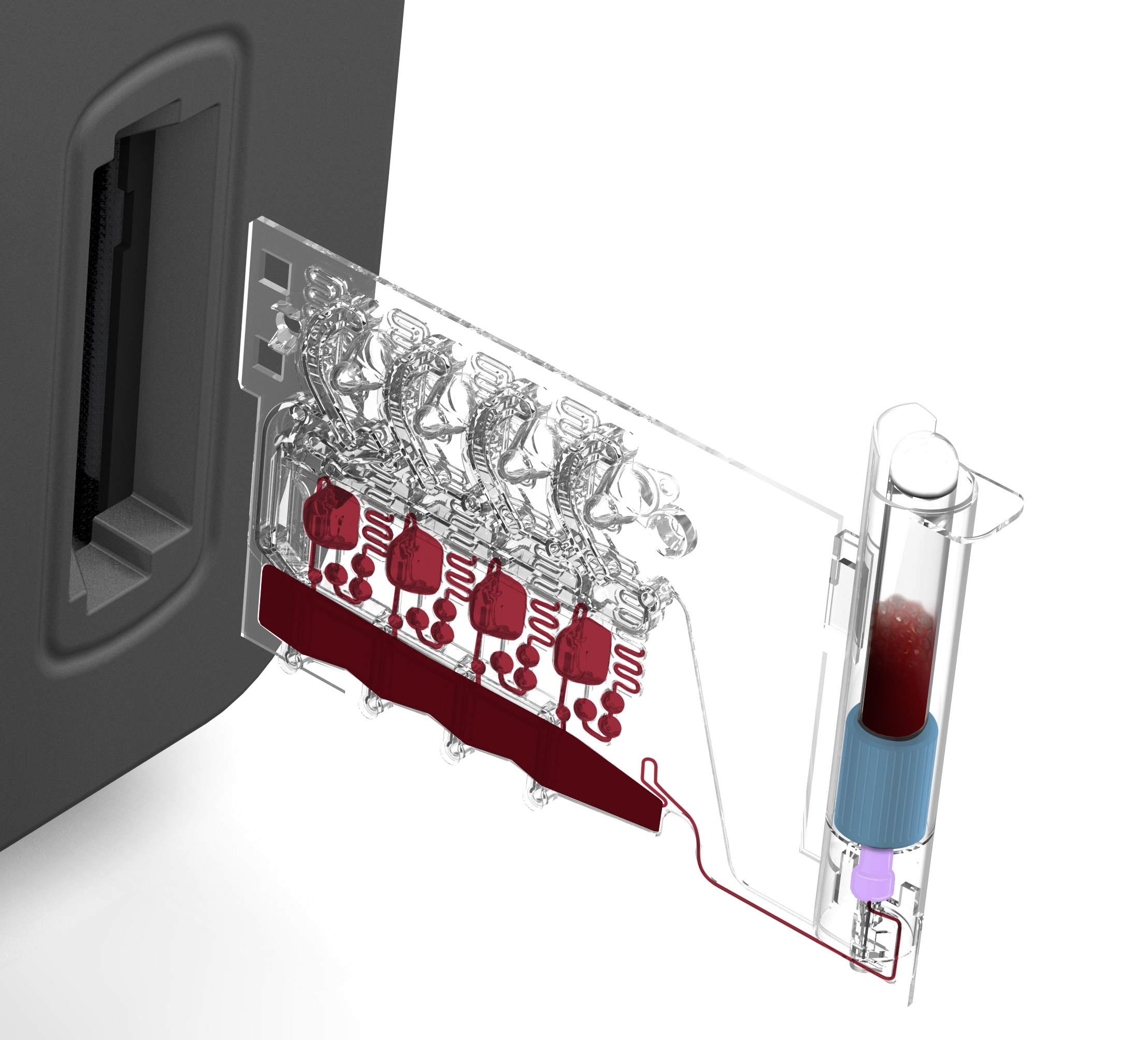
During assay development, reagents are often created on demand for each test. For a product to be successful, however, reagents must withstand uncontrolled shipping environments and long shelf lives. While refrigeration and cold shipping are available options, they drive up disposable costs and are unappealing for facilities with limited refrigerator space. Dried reagent formats are another popular option that can extend the life of otherwise unstable wet reagents, but not all reagents are conducive to being lyophilized, pelleted, or dried-down. Furthermore, dry reagent storage comes with its own challenges of managing moisture and humidity.
While every assay is different and will lend itself to a different reagent storage solution, best practice would be to minimize the number of reagent storage formats. Notably, the storage of both dry and wet reagent types is a common challenge, requiring the use of foil barriers to reduce vapor transmission between liquids and lyos, powders, or dried reagents. Communication between design and assay teams during early product architecture definition is critical to choosing reagent storage solutions that both allow for successful assay integration and minimize design complexity.
While there are many considerations to examine in developing a product system to perform your assay, realistic assay requirements and reagent storage formats are among the most critical to identify early in the product development process. Let us know how Key Tech can support your transition from benchtop to market!
- Working with Client Assay Teams - May 7, 2020
- The Rise of Wearables: How to design a good one - April 2, 2019
- How to Nurture Today’s Young Engineer - March 10, 2014

