18 Nov Key Tech Pharma Process, Phase 2 – Approach to De-Risking Concepts for Architecture Freeze
Drug delivery devices are becoming smaller and more complex, with increased user interactions and advanced technology integration—which also makes these products more exciting and challenging to build.
The first phase of designing a new device involves brainstorming, concept development, and early vetting. The product development team then selects a design concept to further explore, through the process of “de-risking.” In this second phase of design, the team conducts a set of activities that takes a deeper look at the particulars of the design to test its viability. The evaluation of specific risks will hopefully lead to improvements in the design that will reduce or eliminate these risks. With de-risking, the team is still trying to refine the architecture and production directions, vet further design solutions, and prove viability. Will the design hold up against the various technical and usability challenges the team will address? The ultimate goal is to de-risk the design concept to the point where the team has enough confidence to freeze device architecture and start thinking about commercialization.
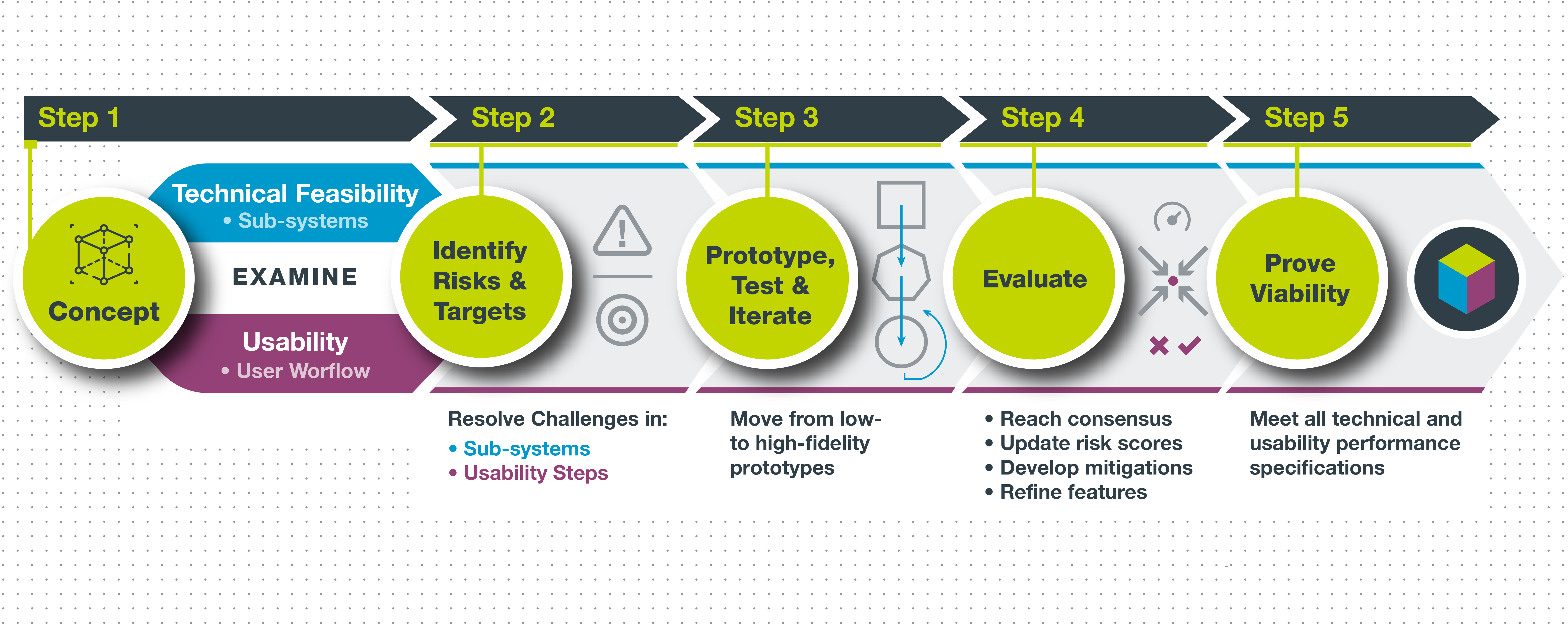
Using our deep experience in designing pharma solutions, Key Tech has developed a five-step approach to de-risking that breaks the concept down into its functional elements and evaluates the performance of the design concept across these de-coupled elements, using rapid prototyping, testing cycles, and other tools. De-risking ultimately reveals the relationships between the critical design parameters and the user experience, which are hopefully strong enough to move the project forward.
Step 1. Break the concept down into functional blocks (technical and usability).
During de-risking, we focus on two critical aspects of the device—technical feasibility and usability.
For technical feasibility, we analyze the critical functionalities of the device by breaking them down into sub-systems that can be studied in detail. Examples of sub-systems include plunger actuation, container piercing and sealing, and fluidic circuits.
For usability, we consider the critical steps in the user workflow and how the cues and design language on the device impact user actions and define the required use steps. Some examples are drug and device inspection, container loading, actuator trigger, and dose completion.
Step 2: Identify risks and performance targets in each sub-system.
For each subsystem, risks and requirements can be identified and targeted. In a plunger actuator sub-system, for example, risks might be actuation force and speed, actuator alignment and position accuracy, power consumption, and miniaturization.
The goal in Step 2 is to highlight the top challenges for each sub-system or usability step that needs to be evaluated and resolved before concept success and product viability can be claimed. Risks where the concern level is high, or where the solution is fundamental to the device architecture or user experience, take the greatest priority and are addressed first. It is critical in this stage to collect input from all relevant stakeholders to understand and prioritize the various risks and acceptable performance targets.
Step 3. Develop prototyping and test plans to assess performance along a timeline of increasing prototype fidelity cycles, with the flexibility to iterate as needed.
Now that the concept risks and challenges have been resolved, the next step is arranging the concept prototyping, testing, and evaluation cycles on a schedule that allows for engineering, human factors, and stakeholder teams to work in parallel; this maximizes time efficiency, collaboration, and exchange.
The beginning of the timeline should start with the highest technical-risk items, or those key sub-systems that will require multiple rounds of user evaluation.
The fidelity of prototypes at the beginning of the timeline should be low, such as engineering benchtop parts and fixtures and usability form and function mock-ups. The goal with these low-fidelity prototypes is to assess first principles of function and user experience. Findings from these early evaluations can then be used to improve the design.
Where appropriate, thorough test plans should be developed and scheduled for complex technical features or demanding performance requirements.
About halfway through the timeline, the development team uses the results from initial experiments and use studies to refine the concept and build more advanced prototypes that combine features and functions. The goal in Step 3 is to evaluate how these combined elements work together and to start identifying optimization constraints.
Prototypes become high fidelity, with full technical functionality and user-facing design features, toward the end of the timeline. These prototypes allow for both engineering performance testing and/or simulated use study, depending on the risks being addressed.
Step 4. Reach consensus on evaluation findings to update concept risk scores, develop mitigations, and refine concept features for next cycle.
Throughout the de-risking process, it is essential to regularly bring the stakeholder teams together to discuss evaluation results and collaboratively determine the next steps. Some evaluations may be straightforward and clearly show that a particular solution is successful, with low-risk levels; others may require down-selection discussions or even new cycles of re-design and evaluation to address design concerns.
The goal in Step 4 is to determine what features are working and are ready to mature through further development and which features need continued de-risking activities. Objectives for additional de-risking activities should be defined, with updated timelines and test plans.
This rigorous process allows the concept to organically evolve into an optimal solution, supported by data that takes the design to the third and final phase—scale-up and commercialization.
Step 5. Demonstrate concept viability with a high-fidelity prototype that can perform full workflow for engineering performance testing and simulated use evaluations.
The last step in the de-risking process is demonstrating that all technical and usability performance specifications can be met, without unacceptable use errors.
Achieving this requires the highest-fidelity prototypes to simulate the full workflow experience of a commercial device, so that all user behaviors and device mechanics can be accurately assessed—this includes packaging, labeling, instructions for use, hardware, and software.
The materials and manufacturing methods selected should balance the associated cost, time, performance, and appearance requirements brought forth in the evaluations.
For simulated use studies, participants should be recruited using an appropriate sample size and population strategy, to cover all potential use cases and yield robust findings
For engineering performance testing, sample sizes and test protocols should be determined so that findings are reliable and can be used as part of the design history for subsequent commercial design phases.
Conclusion
Key Tech has employed our five-step de-risking approach on numerous complex pharmaceutical device projects, optimizing product development, and shortening the commercialization process. Key to this success is the close collaboration with our clients and engaging technical, usability, and stakeholder evaluation activities simultaneously. Our ability to use our engineering experience from outside the pharmaceutical industry often results in more creative prototyping and evaluation strategies. This is the most efficient way to build confidence in the product architecture, avoiding costly re-work later in development.
The third and final installment in this series is entitled “Phase 3: Design for Scale.” It will describe our approach to transitioning a design from its proven concept state (the final output of Phase 2 architecture de-risking) into a design more suitable to mass production and commercialization, while under regulatory design controls. Primary considerations will be balancing cost, design for manufacturing and assembly methods, regulatory requirements and testing, and contract manufacturer partnerships.
- Key Tech Pharma Process, Phase 2 – Approach to De-Risking Concepts for Architecture Freeze - November 18, 2020
- How to Invent Novel Pharma Solutions—Phase 1 - June 4, 2020
- Designing Complex Medical Fluidics Systems - January 8, 2020

