04 Jun How to Invent Novel Pharma Solutions—Phase 1
Key Tech collaborates with pharmaceutical companies around the world to develop innovative solutions for some of their toughest drug-delivery challenges. Our engineers especially enjoy solving design problems where creative and scientific thinking need to work together to uncover the optimal solution.
The foundation of the Key Tech approach that allows for true innovation is understanding every aspect of the drug journey. Start with the big picture that focuses on real-world scenarios. For example, how and where will the drug be used? Who is the user? What experience level is required? How critical or sensitive is the product—is it a high-alert, emergency-use drug or part of a routine procedure? Does the drug require environmental/temperature controls? It is also critical to know about frequency/speed: how often is the product used? How important is quick preparation of delivery?
This holistic perspective toward all aspects of the drug journey, and how they inter-relate at each step of the drug cycle, is necessary before exploring detailed technology solutions. There is no strict “process” of evaluation, since each project challenge is unique, with different goals and expectations. Instead, a comprehensive holistic overview fosters deep understanding of the problem, achieves inspiration from creative thinking, and encourages design and iteration to prove and refine ideas along the way—ultimately resulting in a solution that meets or exceeds expectations.
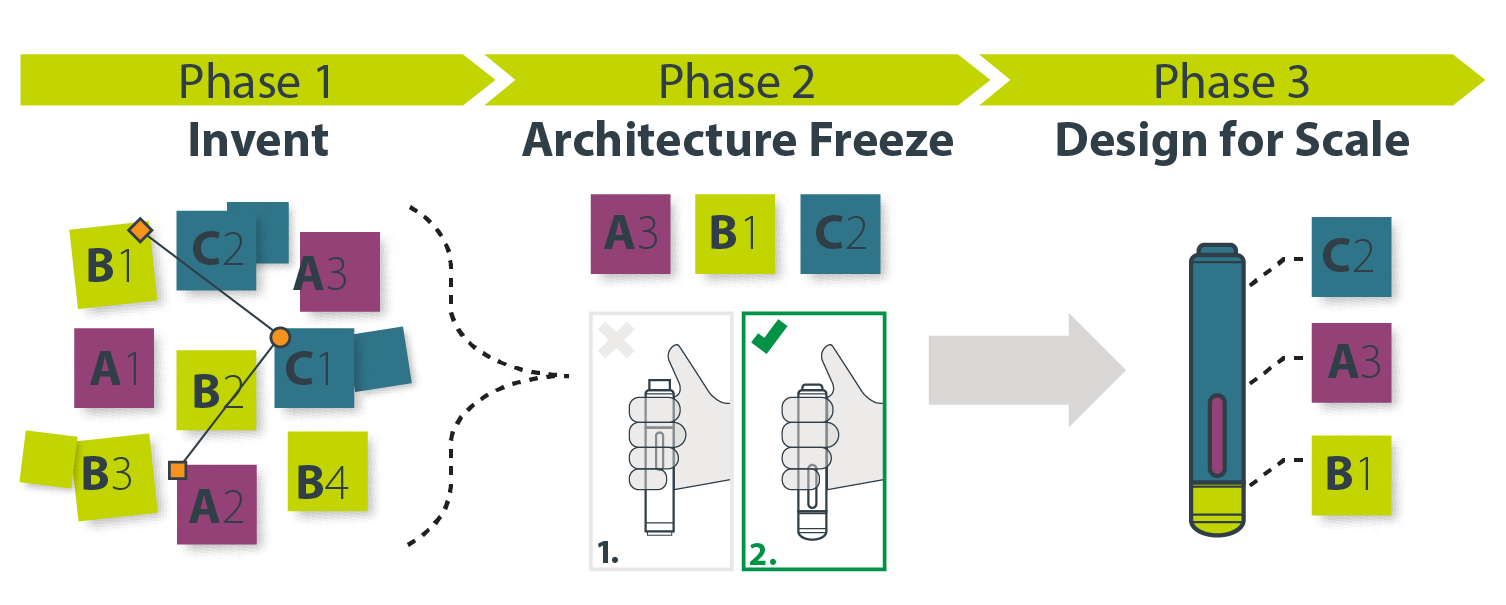
We define this holistic concept generation and refinement work as ‘Phase 1’ in a Three-Phase process of bringing a new drug delivery product to market, which consists of eight different parts or steps:
1. Understand the drug journey.
A “drug journey” entails how a drug product moves from the factory where it is made, through packaging, storage, distribution/sales, and finally to where it is used. This detailed evaluation of the drug’s journey is a must for helping the client and the design team understand relevant trends, pain points, and opportunities for adding value for the patient and the client.

- Break down the drug journey.
The drug journey is often further separated into “use steps”—actions the ‘user’ must take to access and implement the drug or device. The ‘user’ is the person responsible for giving the drug to the patient, which can include cases of self-administration. This knowledge helps us to brainstorm the ideal user experience. The individual use steps can then be organized into a workflow map. We’ve seen cases where some OEMs do not fully understand the use cases and drug journeys for their own products.
- Brainstorm possible high-level technology solutions.
Solutions to each ‘use-step’ are explored, focusing on technologies/methods that can best achieve the task. It is often helpful to study processes developed for other markets, such as in-vitro diagnostics, therapeutic devices, and even consumer products. This is best accomplished using a multidisciplinary design team, which brings a fresh perspective to the challenge. We regularly include ‘users’ to our brainstorming sessions, as well as anyone that plays a role in the drug journey.
- Synthesis—putting the design concept together.
Brainstorming results are grouped to form families of solutions that have common themes, such as durable versus disposable, in-home versus clinic, manual versus semi-automated, primary containers versus custom containers, and a variety of label and packaging options. Each use-step must be addressed in the synthesis. Simple solutions, which provide low unit cost, manufacturing simplicity, and performance reliability, are almost always the leading design goals with pharmaceutical devices, but don’t down-select too quickly. Nuggets of value will likely be found by developing a full range of solutions.
- Embody concepts.
Here we begin to shape the solution space into high-level concepts that combine use and technical features into end-to-end solutions. These are called ‘high level’ because not every detail of every feature is known, but we can assess which concepts have promising potential. The design team imagines the different concepts in use, compares the advantages and disadvantages, and starts to evaluate the best ideas.
Other considerations for the vetting process are high-level business implications, , hospital storage, in-home use, and packaging and kitting requirements.
- Compare and contrast.
The design team looks for opportunities to further refine the remaining concepts or designs and troubleshoot any gaps or possible weaknesses that were identified during early vetting. The high-level concepts are placed in a comparison table and scored according to various metrics. Top metrics include cost, design complexity, ease of use, supply chain risk, and container consideration.
- Choose the best concept for further development and de-risking.
After going through the drug journey, brainstorming, concept development, and early vetting, the team selects the leading idea(s) that score the best across all metrics. Product development then moves on to “de-risking”—performing a set of activities that helps build confidence that the idea will really work. This includes evaluating specific risks that can hopefully be reduced or eliminated through advanced design and evaluation activities. Findings are organized in a table for ranking.
In reading through the above, it’s worth emphasizing that the development of the real physical prototypes has not yet begun. We are still trying to define and refine an architecture and production directions. These eight steps are only the first phase of developing a novel pharma solution. The next blog will discuss Phase 2—how to further improve your vetted and de-risked designs, prove them viable, and get them into production.
What is your challenge? Reach out to discuss it with our experts at TalkToUs@keytechinc.com
- Key Tech Pharma Process, Phase 2 – Approach to De-Risking Concepts for Architecture Freeze - November 18, 2020
- How to Invent Novel Pharma Solutions—Phase 1 - June 4, 2020
- Designing Complex Medical Fluidics Systems - January 8, 2020

