21 Mar Optics Integration in Tech
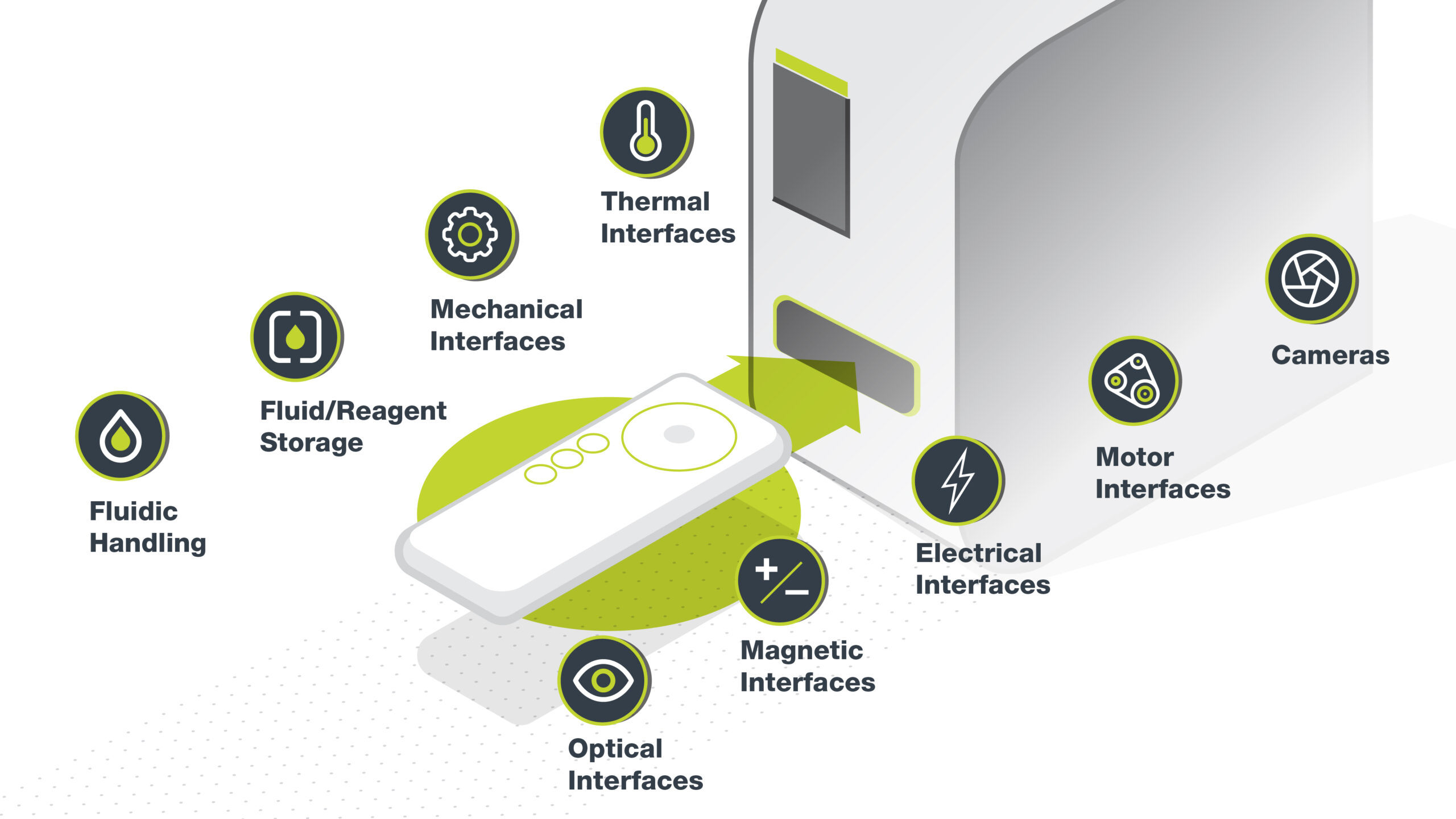
Key Tech has implemented optical solutions in medical and industrial systems for the last 20 years and has experience with both non-imaging (or anidolic) applications and imaging applications (microscopy). Microscopy is the field of optics that uses a microscope to image a sample, and has...