Category
IVDAbout This Project
UltraCrit
Hematocrit POC Full-Scope Development
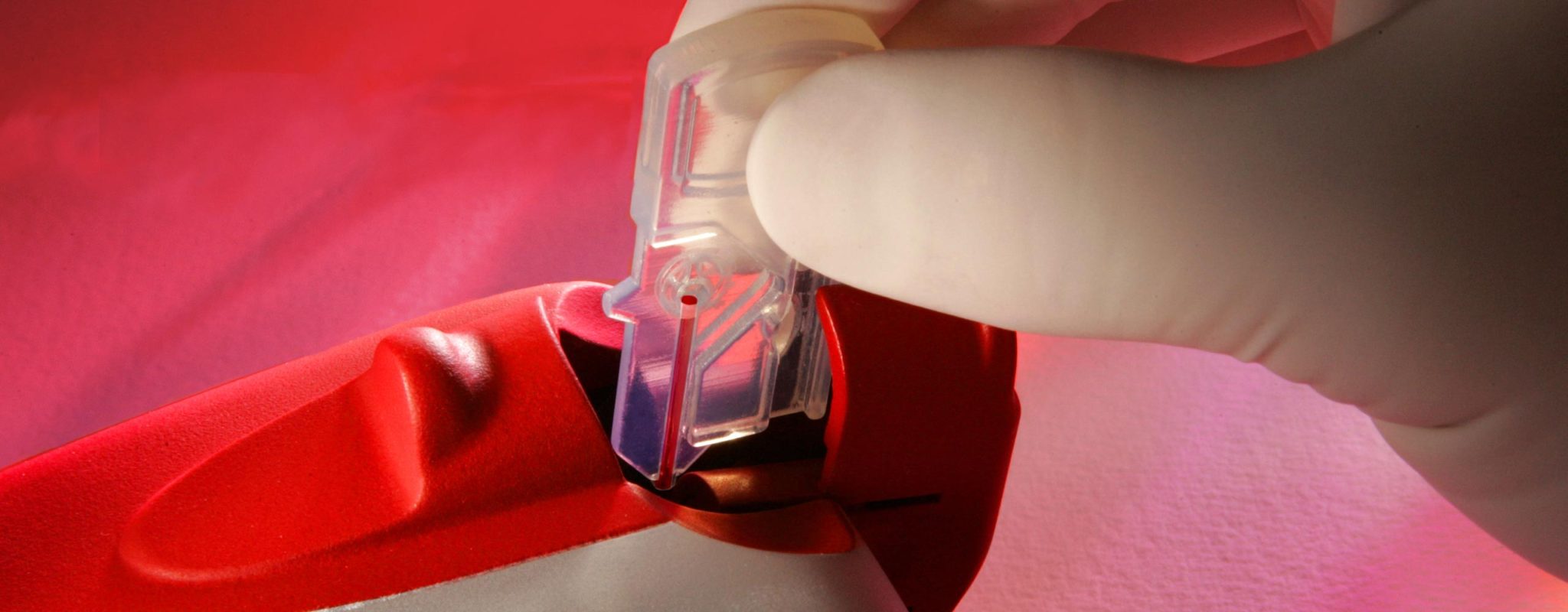
Measuring red blood cells in a hand held device allows blood banks to reject fewer donors, thus increasing the blood supply. The UltraCrit is the first ultrasound point-of-care blood meter. Key Tech developed this CLIA-waived device from concept to market – starting with developing the ultrasound time-of-flight algorithms to detect blood hematocrit. The ultrasound measurement provided the science to achieve greater accuracy.
The sample collection cartridge wicks blood from a finger prick, and understanding the microfluidics of this blood flow resulted in development of a novel shape memory alloy wire pump that generates the vacuum needed to pull the blood into an interrogation region. Key Tech ran pre-clinical trials, verification and validation, and submitted the device for FDA 510k and CLIA waiver. The UltraCrit is now owned and distributed by EKF Diagnostics. Download a detailed Product Development Case Study.
- Ep 45 How We Ran a Clinical Study Without Millions - June 9, 2026
- Ep 44 The Real Cost of Adding Cybersecurity Late in Medical Device Development - February 4, 2026
- Ep 43 MedTech’s 11 Year Exit Problem— and What It Means for Raising Capital - December 12, 2025






